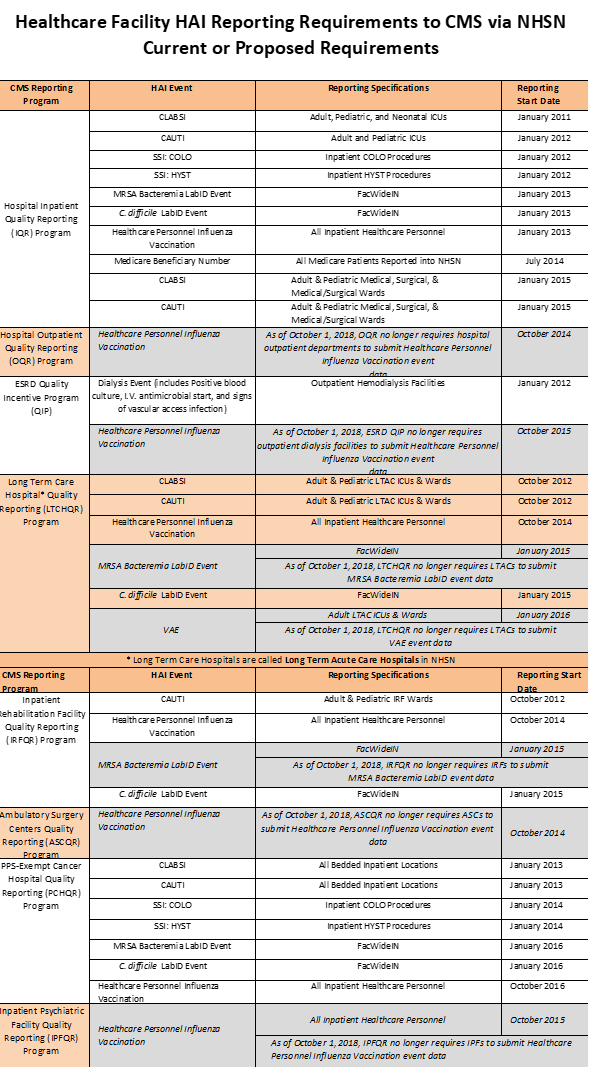
CMS released its final rule for the FY 2011 Hospital Inpatient Prospective Payment System (IPPS) on July 30, 2010. This rule requires that all hospitals participating in the Medicare program report data on specific HAIs through the Centers for Disease Control and Prevention’s (CDC) National Healthcare Safety Network (NHSN). Reporting requirements in this final rule include:
DICON has developed reporting systems for data collected locally that comply with existing and future (i.e. anticipated) mandatory reporting requirements for hospitals. (In other words, we can take care of mandatory reporting for you and give you confidence that your data is accurate and simultaneously benchmarked with a large cohort of similar size hospitals in DICON.) Also, because we have validated our surveillance methods and because we have databases containing information on > 16 million patient days and nearly 1 million surgical procedures, we can help you understand, explain and use your locally collected data. Finally, we can help protect you from unfounded speculations about the meaning of your publically reported data.
